Benefits
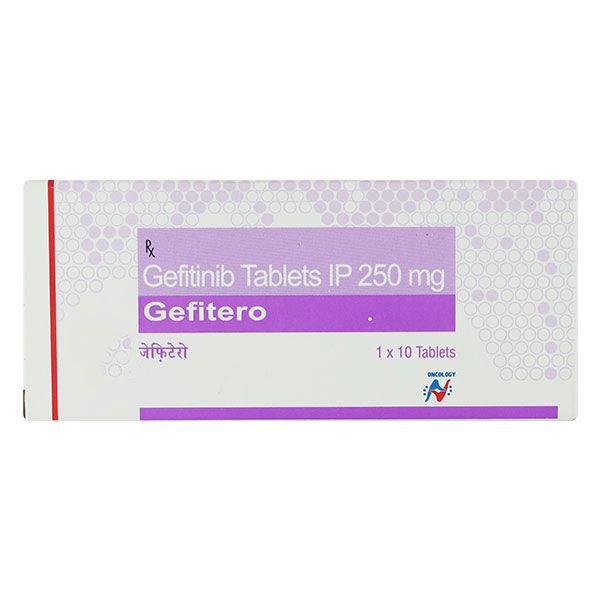
Gefitero Tablet helps treat non-small cell lung cancer. It works for both smokers and non-smokers in that condition. Doctors often give it on its own or mix it with other medications to fight the disease. This is a powerful type of cancer drug, so you really need to go over the risks and benefits with your doctor before starting. During the whole treatment period, stay away from alcohol and keep yourself well hydrated by drinking enough fluids.









Reviews
There are no reviews yet.